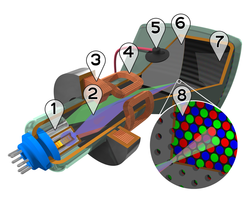
Cutaway rendering of a color CRT:
1. Three Electron guns (for red, green, and blue phosphor dots)
2. Electron beams
3. Focusing coils
4. Deflection coils
5. Anode connection
6. Mask for separating beams for red, green, and blue part of displayed image
7. Phosphor layer with red, green, and blue zones
8. Close-up of the phosphor-coated inner side of the screen
1. Three Electron guns (for red, green, and blue phosphor dots)
2. Electron beams
3. Focusing coils
4. Deflection coils
5. Anode connection
6. Mask for separating beams for red, green, and blue part of displayed image
7. Phosphor layer with red, green, and blue zones
8. Close-up of the phosphor-coated inner side of the screen
The cathode ray tube is a vacuum tube containing a source of electrons, like an electron gun, it also has a fluorescent screen, with internal or external means to accelerate and deflect the electron beam. The cathode ray tube uses an evacuated glass envelope which is large, deep, heavy, and relatively fragile. The cathode ray is a stream of electrons in a vacuumed tube. An evacuated glass tube, equipped with at least two metal electrodes has a voltage applied to it. The voltage is a cathode or negative electrode and an anode or positive electrode. The cathode ray was first observed in 1869 by Johann Hittorf, a German physicist, but was named in 1876 by Eugene Goldstein kathodenstrahlen, or cathode rays. Electrons were first discovered using the cathode ray by J.J. Thomson in 1987. Thomas showed that the rays also had negatively charge particles, which were the electrons. Thomson found that the rays could be deflected by an energetic field. He was able to measure the particle's mass by comparing the deflection of a beam of cathode rays by electrical and magnetic fields. He discovered that they were 2000 times lighter than a hydrogen atom. He concluded that the rays had negatively charged particles.
One of the important things about the discovery of the electron was the electron microscope. It was invented in 1928 by Ernst Ruska. The electron microscope uses a stream of electrons to magnify something. Electrons have small wavelengths so they can be used to magnify objects that are too small to be resolved by light. Ruska used a strong magnetic field in order to focus the electrons into an image in a stream.
For information on how the cathode ray tube is go to: http://www.youtube.com/watch?v=XU8nMKkzbT8
For information on how the cathode ray tube is go to: http://www.youtube.com/watch?v=XU8nMKkzbT8
Sources:
http://www.bookrags.com/research/cathode-ray-tube-woi/
http://dev.physicslab.org/Document.aspx?doctype=3&filename=Magnetism_CathodeRays.xml
http://en.wikipedia.org/wiki/J._J._Thomson#Discovery_of_the_electron
http://en.wikipedia.org/wiki/Cathode_ray_tube
http://en.wikipedia.org/wiki/Cathode_ray
http://www.bookrags.com/research/cathode-ray-tube-woi/
http://dev.physicslab.org/Document.aspx?doctype=3&filename=Magnetism_CathodeRays.xml
http://en.wikipedia.org/wiki/J._J._Thomson#Discovery_of_the_electron
http://en.wikipedia.org/wiki/Cathode_ray_tube
http://en.wikipedia.org/wiki/Cathode_ray
I like how your blog was short, but to the point as well. You provided very interesting information. You could have made your layout a little more interesting by changing the font, making the font larger, adding more color, or changing the spacing. The picture and video were very helpful. Great job on your blog!!
ReplyDeleteI really like the information you gave in your blog but I did feel that It was a little too condensed. Perhaps Spacing it out a little would make it easy on the eyes. Usage of bullet point or even double spacing the paragraphs would make the blog post easier to read.
ReplyDeleteFantastic blog post Daniel! Keep up the good work! Your blog post was very informational and relatable but could have used more pictures. After you're done with blog posts fuel up with a great bowl of frosted flakes, therrrrrr GREAAAAT!
ReplyDeleteYou did a really good job on picking out important information and higlighting on the main points. The picture was also really helpful but it would have been nice to add more pictures. It would have been easier to follow if you had organized your information into different sections. You could also have expanded a little more on the experiment of the cathode ray, but overall you did a really good job.
ReplyDelete-Michelle